Orignal Author
Yi-Heng Sung
English Translator
Yi-Heng Sung
Copyright© LIS情境科學教材

Water is the origin of life and is vital for all living beings. From ancient times, water has been a focal topic. Ancient Chinese philosopher Laozi said, “Water can carry a boat (水能載舟)” alluding to buoyancy: an object sinks if its density is greater than the liquid’s and floats if it is less dense. Similarly, the Taiwanese band Mayday quipped in a song, “Water can also cook porridge (亦能煮粥),” emphasizing water’s long-standing role in cooking. However, the connection between fluids and heat only began to be understood in the late 18th century.
Born in 1753, Benjamin Thompson (1753–1814), later known as Count Rumford, grew up in the Massachusetts colony of New England. As the only child of a farming family, he displayed an early passion for science experiments. After finishing school at 13, he worked various jobs—apprentice, clerk, and teacher—while pursuing his scientific interests. He even attended Harvard science lectures. Despite his aspirations, Thompson led a modest life until 1772 when he met Colonel Timothy Walker, one of the key figures in his life.
Walker invited Thompson to teach in Rumford, where he met Walker’s wealthy daughter, Sarah. Though 14 years older and recently widowed, Sarah fell for Thompson, and they married within four months. This marriage elevated Thompson’s status, making him a landowner and researcher. His connections with the colonial governor led to his appointment as a major in 1773.
When the American Revolutionary War broke out in 1775, Thompson supported the British. His assistance to British forces made him a target of local hostility, forcing him to flee his home in 1776 and relocate to England with the retreating British army, sadly, leaving his family behind.


In England, Thompson leveraged his military connections, gaining the patronage of George Germain, Secretary of the Colonies. His military career advanced, yet he never lost his passion for science. While working at the Colonial Office in London, he also cultivated relationships with members of the British scientific community of his time. Thompson held various positions, rising from the rank of major to colonel, and retired from military service at the age of 30. Post-retirement, Thompson toured Europe, eventually earning the favor of Karl Theodor, Elector of Bavaria. The Elector held Thompson in high regard and appointed him to many key positions, steadily elevating Rumford’s status in the Kingdom of Bavaria. Despite his rise to the upper echelons of society, Rumford remained devoted to his scientific pursuits. Driven by an experimental spirit, he applied scientific methods to improve many lives, such as developing efficient stoves and nutritious soups for plague-stricken populations. His contributions earned him the title of Count Rumford in 1793. He chose “Rumford,” the name of the place where he first rose to prominence, as his noble title.

On a sunny day, Thompson used an old, poorly stored thermometer with a spherical base over 10 cm in diameter. This thermometer hadn’t been used for a long time and was poorly maintained, so the liquid inside was not clean, with dust particles floating in it. Even so, because the liquid was colored, the dust wasn’t particularly noticeable at first glance. However, after Thompson heated the thermometer and left it by the window to cool down, sunlight passed through the thermometer, making the dust inside the liquid highly visible. To his surprise, he observed the dust moving rapidly and in a very orderly pattern: an upward current in the center and a downward current along the sides (as illustrated below).
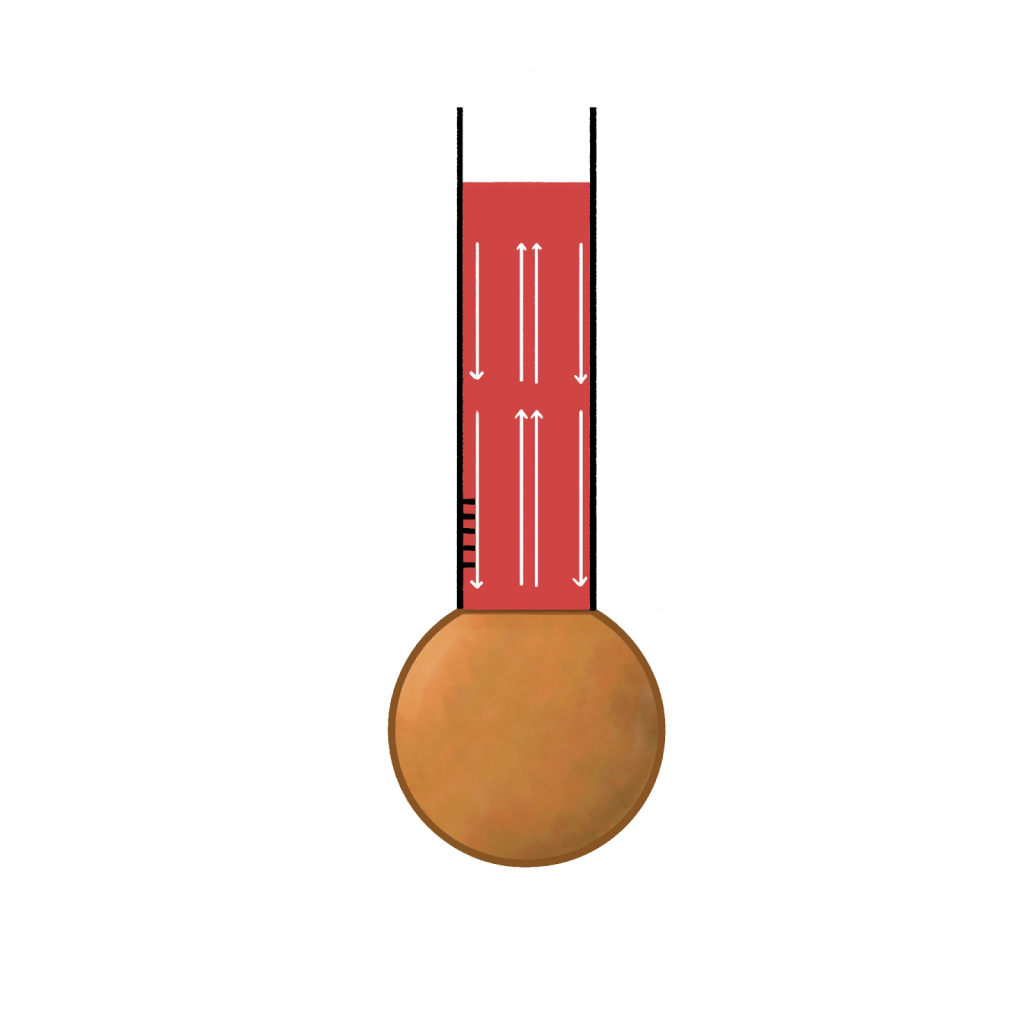
Fascinated by this phenomenon, Thompson created an experimental device to visualize the flow more clearly. He replaced the dust with amber particles, which had almost the same density as the saline solution, so the particles could neither sink nor float. By this method, he could create water flow by changing the temperature, and the amber particles suspended in the liquid would move along with the flow. This allowed him to clearly see the upward flow in the center and the downward flows on both sides that Thompson had previously observed. Since the amber particle shared the same density as the solution, the particles would sink if they became denser; the particles would float on the surface if they became less dense. Thompson connected this observation to the principles of density and buoyancy: objects with greater density sink, while those with lower density rise. He also noticed that in a stationary thermometer, internal flow appeared only during cooling from a high temperature and stopped once the temperature reached equilibrium with the surrounding room. This led him to hypothesize that changes in temperature might cause changes in the liquid’s density. This implied that when the liquid’s temperature changed, the resulting density differences could cause the liquid to rise and fall, creating the observed flow. Thompson hypothesized that liquid motion resulted from local temperature variations, the warmer, less dense liquid would rise, forming upward currents; while the cooler, denser liquid would sink, forming downward currents. Once the temperature of the liquid became uniform, its density would also become uniform, and the flow would cease. This insight laid the groundwork for understanding heat convection, another type of heat transfer than conduction.
Beyond water, fire has always been a trusted companion of humanity. Over a million years ago, our ancestors already knew how to use fire, marking an essential milestone in the development of human technology. From centuries of experience with heating, humans observed consistent patterns in how heat transfers. Heat always flows from areas of higher temperature to lower temperature, spreading outward evenly in all directions, as known as heat conduction. For example, when cooking a steak, the side of the meat closest to the pan heats up and cooks first. The heat gradually moves from the bottom of the steak to the top, which is farther from the heat source.
After Thompson discovered the cause of the current in the thermometer, he continued conducting experiments about fluids and heat. During one of the experiments, He found another interesting fact! Thompson compared the ice melting rate of ice placed at the surface and bottom of a container filled with boiling water. While the surface ice melted in 3 minutes, the bottom ice took over 2 hours to melt halfway. This drastic difference contradicted the understanding of heat conduction at the time, which should have melted both equally. The temperature of the hot water in the container is uniform, and the ice cubes are of the same size. Since the heat transfer should occur from water at the same high temperature to ice cubes at the same low temperature, the melting speed should be identical. Therefore, Thompson speculated that there might be a way heat propagates beyond just conduction.
Thompson reflected on his earlier experiments with internal fluid flow in thermometers. When the temperature rises, the liquid’s density decreases, creating upward currents. This means that the rising liquid, due to its lower density, is also the heated portion of the fluid. Thompson theorized that liquids might transfer heat through internal flow. As the less dense, warmer liquid rises, it carries heat to the upper layers, making the liquid at the top warmer than the part at the bottom. When an ice cube is placed on the surface of hot water, the colder liquid near the ice sinks, and warmer liquid from below rises to continue melting the ice. Conversely, if the ice cube is placed at the bottom of the container, the water near the ice cools and its density increases, causing it to stay at the bottom. Meanwhile, the warmer, less dense water remains at the top. This lack of downward movement of hot water and the persistent cold water at the bottom result in slower melting of the ice cube at the bottom.
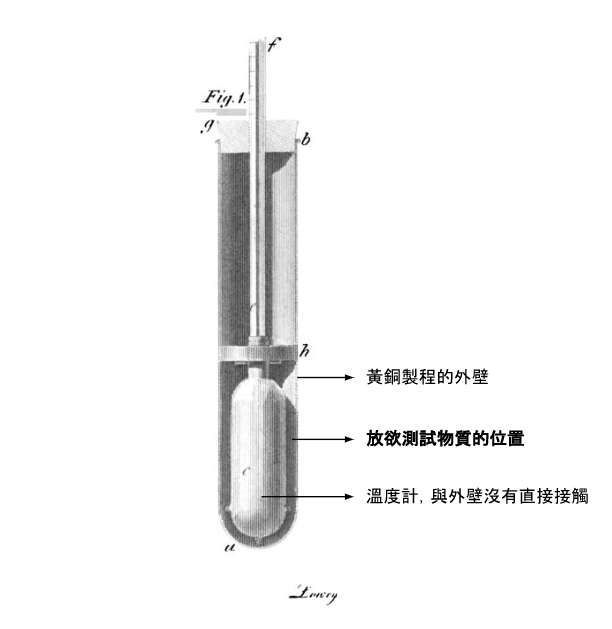
If the hypothesis was correct—that heat in liquids is transferred through internal convection—then blocking the fluid’s internal motion should slow heat transfer. He therefore devised an experimental apparatus with a thermometer at its center, surrounded by a brass casing without direct contact. The space in between could be filled with different materials.
First, Thompson submerged the entire setup in ice water until the thermometer read 32°F. Then, he placed different experimental materials between the thermometer and the brass casing and immersed the setup in boiling water. He recorded the time it took for the thermometer to rise from 40°F to 60°F. By comparing the heat transfer rates of different materials, Thompson could determine whether fluid motion affected heat transfer. Using mashed apples as a viscous medium, Thompson observed slower heat transfer compared to water. He concluded that the fibers in the mashed apples hindered the internal fluid movement, slowing heat transfer.
To further test his hypothesis, he adjusted the fiber content in experimental solutions. Thompson measured the fiber content in mashed apples and replicated this composition using water mixed with down feathers. Testing this solution in his apparatus showed that the time required for heat transfer matched that of mashed apples, confirming that the down impeded internal fluid flow. He then created solutions with varying concentrations of feathers and observed that as the feather content decreased, the time for the thermometer to rise from 40°F to 60°F also decreased. This demonstrated that reducing the obstruction to fluid motion increased the speed of heat transfer.
Thompson’s discovery of fluid motion and heat transfer forms the basis of what we now call convection. Though the term was introduced later in 1834 by chemist William Prout, Thompson’s experiments revolutionized the understanding of heat flow in nature and engineering.
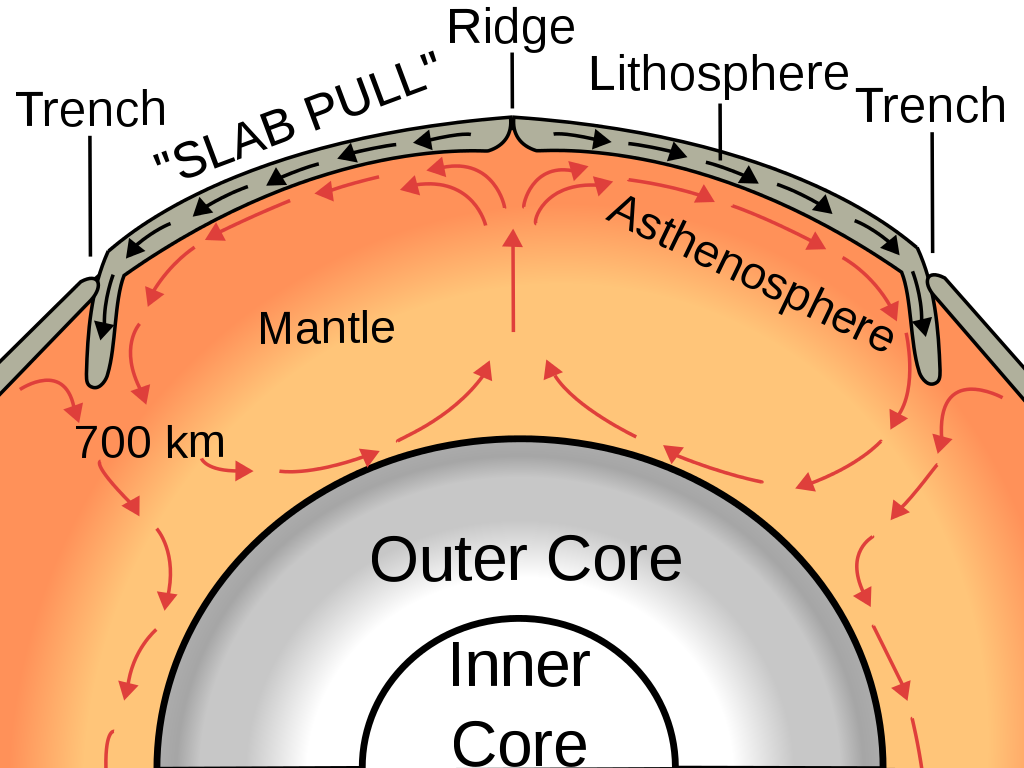
Convection is a common occurrence in nature, and Thompson’s discovery of its principles has significantly contributed to the development of many scientific fields. For example, ocean currents are driven by convection. Since gases are also considered fluids, this discovery has profoundly influenced the study of atmospheric science and meteorology. Additionally, the Earth’s mantle, filled with molten rock due to intense heat, exhibits convection (as illustrated below). Understanding convection has helped scientists gain deeper insights into the Earth’s internal processes, proving invaluable in the field of geology.
Beyond its applications in scientific research, understanding convection has also brought undeniable benefits to our daily lives. For instance, cold air is denser and tends to sink, while warm air, being lighter, rises. This is why air conditioners are typically installed near the ceiling—cold air descends and cools the entire room. Conversely, heaters are placed near the floor so that the rising warm air can be distributed evenly throughout the space.
Thompson’s curiosity and dedication to science led him to uncover the phenomenon of heat convection, a process long present in nature but previously unnoticed. This discovery has been foundational for advancements in various scientific disciplines and has improved the quality of human life. While convection may not be the most famous of Thompson’s scientific achievements, it is undoubtedly one of the most impactful.
Got a thought, a project, or just want to say hi? I’d love to hear from you!